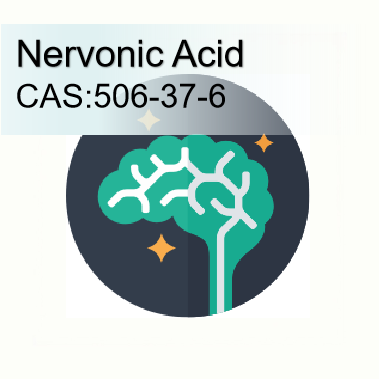
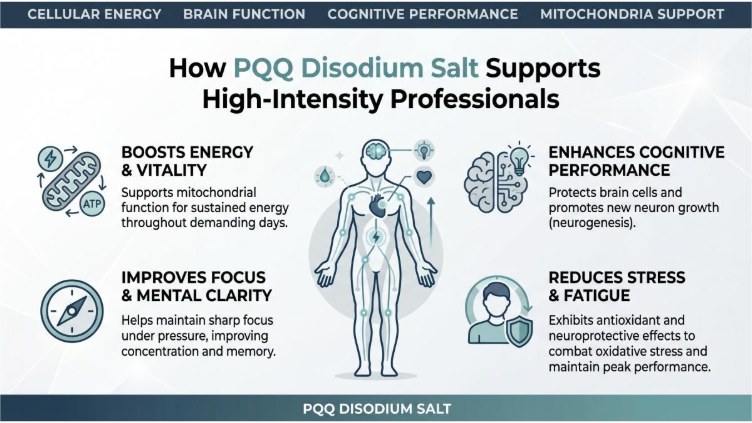
How PQQ Disodium Salt Support High-Intensity Professionals
In the knowledge economy, high-intensity mental output is the defining currency of professional success. Software architects designing microservice architectures, copywriters editing high-converting brand messages, and product visualizers aligning complex UI designs operate in a state of continuous cognitive strain. Yet, many professionals are all too familiar with the dreaded afternoon crash—the feeling of “brain lag” where ideas seem trapped behind a wall of mental inertia. Before diving deeper, many buyers begin their journey by reviewing pqq bulk supplements review resources to understand which products actually deliver on their promises. This slow cognitive processing, commonly referred to as “brain fog,” is not a sign of low motivation; rather, it indicates a critical decline in neuronal metabolic efficiency. For international supplement brands, private label manufacturers, and distributors, what is pyrroloquinoline quinone is a question they ask when first encountering this unique compound—and the answer lies in its rare ability to energize neurons at the cellular level. PQQ Disodium Salt has emerged as a key nootropic ingredient to revitalize cerebral energy and eliminate mental cognitive sluggishness.
PQQ Disodium Salt: The Cell-Level Fuel for Your Neural Circuitry
Methoxatin disodium salt, another name of PQQ CAS No.122628-50-6, works as a powerful cognitive enhancer by supporting metabolic pathways in cerebral neurons. PQQ is one of the few nutritional molecules capable of passing through the blood-brain barrier (BBB) with high bioavailability.
Once in the brain, PQQ disodium salt works to improve cognitive performance in two main ways:
·It stimulates the endogenous production of Nerve Growth Factor (NGF). Research has shown that PQQ promotes NGF synthesis in astroglial cells without cytotoxicity—in fact, it demonstrated the strongest NGF-promoting activity among the coenzymes tested. NGF is essential for brain health, supporting the growth, maintenance, and structural flexibility of vital learning-associated neurons.
·It protects neuronal pathways from oxidative stress and toxic proteins by preventing the accumulation of reactive oxygen species in brain tissue.
By boosting both structural support (NGF) and immediate energy production (mitochondrial ATP), PQQ disodium salt provides comprehensive cognitive optimization.
Clinical Benchmark & Performance Comparison (Without Tables)
The cognitive and protective effects of PQQ Disodium Salt are supported by clear biochemical and clinical study results. Formulation designers can review the following key comparisons:
Nerve Growth Factor (NGF) Synthesis Acceleration: Traditional herbal brain boosters (e.g., standard Ginkgo Biloba or Ginseng extracts) show marginal or indirect support for neural growth factors. In contrast, cell culture studies demonstrate that PQQ can significantly increase the secretion of endogenous NGF in astroglial cells, powerfully aiding structural neural plasticity. Clinical investigations further confirm that PQQ enhances mitochondrial activation, growth, and repair of nerve cells by increasing NGF and NGF receptor expression.
Cognitive Processing & Error-Rate Decline: A randomized, double-blind, placebo-controlled clinical trial evaluating healthy adults who took 20 mg of PQQ disodium salt daily for 12 consecutive weeks revealed improvements in both general memory, verbal memory, working memory, and attention. A separate 2024 review published in the Journal of Medical Investigation concluded that PQQ exerts antioxidant, neuroprotective, and mitochondrial biogenesis effects, with co-administration of PQQ and coenzyme Q10 further enhancing cognitive outcomes.
Neurovascular Blood Flow Elevation: Multi-channel brain imaging shows that a 20 mg daily serving of PQQ Disodium Salt can significantly increase capillary blood flow to the prefrontal cortex, providing a steady supply of oxygen and glucose to power demanding mental work. PQQ has also been shown to improve cognitive flexibility and executive speed, with younger adults (20–40 years) experiencing faster improvements in cognitive flexibility and executive speed by 8 weeks compared to older populations.
Eliminating the Brain Slump: Formulating the Ultimate Digital Work Blend
For buyers looking to launch products targeting programmers, creatives, and knowledge workers, PQQ Disodium Salt is an ideal primary active ingredient. Developing a dedicated neuro-focus tablet or capsule utilizing 20 mg of PQQ Disodium Salt, paired with 150 mg of standard Bacopa Monnieri, 200 mg of L-Theanine, and 100 mg of Phosphatidylserine, is highly effective.
This synergistic blend supports multiple cognitive pathways, helping to improve memory recall, promote mental calm, and provide clean, non-jittery energy. This premium formulation allows professional users to maintain peak mental focus and avoid the mid-afternoon cognitive slump.
If you are exploring pqq bulk supplements review and wholesale pqq bulk supplements options for your next product line, partnering with a specialized manufacturer ensures you receive high-purity, GMP-certified material that meets international regulatory standards.
LeadingNutra supplier Ingredient PQQ
LeadingNutra your Trusted Partner in Premium PQQ Disodium Salt Manufacturing . [Contact us] today to request a sample or discuss your private label needs.
Written by Market Director ----Jony Tang
[1] Tamakoshi, M., Suzuki, T., Nishihara, E., et al. (2023). Pyrroloquinoline quinone disodium salt improves brain function in both younger and older adults. Food & Function, 14(5), 249 6–2501. DOI: 10.1039/d2fo01515c.
[2] Ikemoto, K., Mohamad Ishak, N. S., & Akagawa, M. (2024). The effects of pyrroloquinoline quinone disodium salt on brain function and physiological processes. The Journal of Medical Investigation, 71(1.2), 23–28. DOI: 10.2152/jmi.71.23.
[3] Shiojima, Y., Takahashi, M., Takahashi, R., et al. (2022). Effect of Dietary Pyrroloquinoline Quinone Disodium Salt on Cognitive Function in Healthy Volunteers: A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Study. Journal of the American Nutrition Association, 41(8), 796–809. DOI: 10.1080/07315724.2021.1962770.
2026.06.06