Coenzyme Q10 in China Health Food Catalog Filing Guide
04/27/2026 10:07:45
The inclusion of Coenzyme Q10 (CoQ10) in the Health Food Raw Material Directory of the People's Republic of China has revolutionized market entry for both domestic and international enterprises. For OEM/ODM partners, the shift from "Registration" —which could take 3–5 years—to "Filing" creates a streamlined pathway to the world's second-largest health food market. However, "simpler" does not mean "easier." This guide deconstructs the technical barriers to entry and the rigorous standards required for a successful filing. Let’s dive into what actually matters for your product’s approval.
CoQ10 Health Food Entry Threshold Understanding China Filing System
The filing system is essentially a transparency and guarantee mechanism. Unlike the registration system, where the efficacy of the product is independently tested by the state, the filing system places the burden of proof entirely on the manufacturer. In other words, you own the responsibility—and the opportunity.
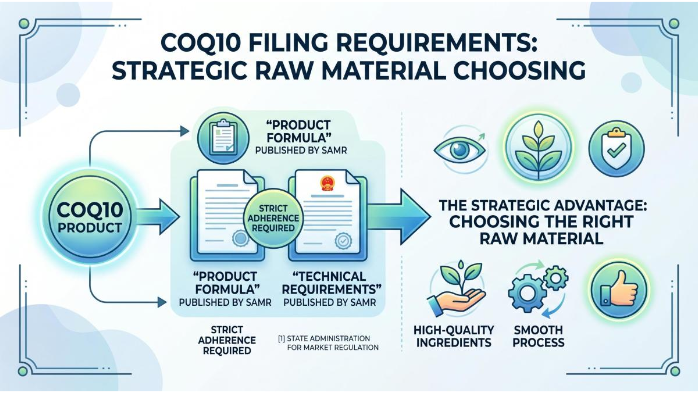
Eligibility for CoQ10 Product Filing
To qualify for filing, a CoQ10 product must strictly adhere to the standardized "Product Formula" and "Technical Requirements" published by the State Administration for Market Regulation (SAMR). This is where choosing the right raw material becomes a strategic advantage.
Single Ingredient vs. Compound Formulas
Under current filing rules, CoQ10 is typically filed as a single-ingredient product. If you wish to combine it with other active ingredients like Vitamin E or Omega-3 for "synergistic effects," you may fall back into the more complex Registration channel. This is a critical strategic decision for OEM clients. Many experienced formulators now prefer Bio-identical Coenzyme Q10 Ingredient because it mimics the body’s natural structure, enhancing absorption and regulatory acceptance—making it ideal for single-ingredient filings.
Comparative Data: Time-to-Market Comparison
Registration Process: 36 – 60 months (Includes human clinical trials and comprehensive state review).
Filing Process: 3 – 6 months (Focused on dossier consistency and raw material verification).
CoQ10 Technical Core Purity Standards and Chemical Profiles
The Chinese regulatory framework for CoQ10 is heavily influenced by the Chinese Pharmacopoeia (ChP) and specific SAMR guidelines for health food materials. Meeting these standards isn’t just about passing the test—it’s about building trust with Chinese consumers.
Co Q10 Purity and Impurity Controls
For a successful filing, the CoQ10 raw material must meet a purity threshold of no less than 98.0%. However, to stand out in a competitive market, many manufacturers are now upgrading to Reduced Coenzyme Q10 98% Purity—the active antioxidant form (Ubiquinol) that delivers superior bioavailability. This higher standard not only satisfies filing requirements but also supports premium product positioning, especially for anti-aging and energy support formulas.
Banned Substances and Synthesis Markers

China strictly prohibits the use of certain chemicals in the CoQ10 manufacturing process. Synthetically derived CoQ10 using "Solanesol" as a base must undergo rigorous testing for residual solvents. The presence of impurities such as "Coenzyme Q9" must be limited based on strict titration limits. That’s why more suppliers are shifting to Microbial Fermented CoQ10 Bulk, which eliminates solvent residues and offers a cleaner, more natural profile that regulators favor.
Purity Benchmarks Comparative
Standard Industry Grade: 95% – 97% (Unsuitable for China Health Food market).
China SAMR Requirement: ≥ 98.0% (Mandatory for Filing).
Recommended Premium Grade: Reduced Coenzyme Q10 98% Purity for enhanced efficacy and filing differentiation.
CoQ10 OEM Client Dossier Documentation and Preparation Guide
For an OEM/ODM factory, providing a "technical dossier" that speaks the language of Chinese regulators is vital. A well-prepared dossier can cut review time by weeks. Remember, the filing officer reads your documents—make their job easy.
The Required Dossier Components
Raw Material Source Documentation: Proof of fermentation sources.
Stability Test Reports: Accelerated stability tests (6 months) and long-term stability data.
Product Safety Assessment: Toxicological evaluation summaries.
When you source Microbial Fermented CoQ10 Bulk, you can provide complete fermentation batch records, which greatly strengthens the dossier’s credibility. Additionally, Sustainable CoQ10 Ingredient Sourcing—such as non-GMO, water-efficient fermentation—is becoming a documented advantage in the filing review process.
Manufacturing Facility Compliance
The factory must hold a "Food Production License" with a scope specifically including Health Food. For international OEMs, GMP (Good Manufacturing Practice) certification is non-negotiable and must be authenticated by Chinese consulates or recognized international bodies. Furthermore, working with a supplier that offers Bulk Ubiquinol for Anti-aging Supplements ensures your finished product meets the highest stability standards required for long-term shelf-life testing.
Avoiding Pitfalls in Co Q10 Market Access with GMP Certified CoQ10 Manufacturer
Many companies fail the filing process not because their product is unsafe, but because of technical inconsistencies in their paperwork. Let’s turn those pitfalls into opportunities.

Labeling and Claims
In China, CoQ10 health foods are permitted to carry specific function claims: "Enhancing Immunity" and "Antioxidant." Any attempt to suggest "Weight Loss" or "Liver Protection" during the filing process will result in immediate rejection. For forms like Bulk Ubiquinol for Anti-aging Supplements, stick strictly to the approved claims—your marketing team can later build creative campaigns around these scientific foundations.
The Importance of the "Technical Product Specification" (TPS)
The TPS is the soul of the filing. It must include the precise excipient list. For instance, the use of beeswax or soybean oil in softgels must meet specific food-grade standards and be listed with exact percentages. When you choose Bio-identical Coenzyme Q10 Ingredient and Sustainable CoQ10 Ingredient Sourcing, these details should be prominently featured in your TPS—they demonstrate technical excellence and supply chain responsibility, two qualities Chinese regulators increasingly value.
Strategic Partnership in a Regulated Market
The Chinese CoQ10 filing system offers a golden opportunity for manufacturers who prioritize technical excellence over shortcuts. By partnering with regulatory experts and maintaining a "Quality-First" approach, B2B companies can ensure their products not only enter the market but thrive within a framework of legal certainty. Whether you opt for Reduced Coenzyme Q10 98% Purity, Microbial Fermented CoQ10 Bulk, or Bulk Ubiquinol for Anti-aging Supplements, the key is to align every ingredient choice with the filing logic.
Leadingnutra the premium supplier for high-purity CoQ10 and Ubiquinol ingredients.Contact us today for expert filing support and sustainable sourcing solutions: lily@leadingchemical.com
[1] China SAMR - Technical Requirements for Coenzyme Q10 Health Food Filing
[2] WHO - Guidelines for Good Manufacturing Practices (GMP) for Medicinal Products
[3] National Institute of Health Food and Drug Control (NIFDC) China
[4] NMPA - Announcement on Further Regulation of Health Food Filing Management (2023)
[5] FAO/WHO - Sustainability Principles for Ingredient Sourcing in Nutraceuticals
English
Japanese
Russian
Korean
गोंगेन हें नांव
Deutsch
Corsu
Guarani
Hausa
Cymraeg
Nederlands
Aymara
Français
Kreyòl ayisyen
čeština
ʻŌlelo Hawaiʻi
डोग्रिड ने दी
ภาษาไทย
հայերեն
فارسی
Hmoob
ދިވެހި
भोजपुरी
繁體中文
Türkçe
हिंदी
беларускі
български
tur
Gaeilge
ગુજરાતી
Magyar
Eesti keel
بالعربية
বাংলা
Azərbaycan
Português
Suid-Afrikaanse Dutch taal
کوردی-سۆرانی
Ελληνικά
español
Frysk
dansk
አማርኛ
Bamanankan
euskara
Italiano
Tiếng Việt
অসমীয়া
català
Suomalainen
Eʋegbe
Hrvatski
Cebuano
Gàidhlig na h-Alba
bosanski
galego




