Spermidine Gains EFSA Novel Food Approval in 2024 Regulatory Update
04/26/2026 15:35:20
The European dietary supplement market, governed by the rigorous European Food Safety Authority (EFSA), represents a high-barrier, high-reward territory for longevity ingredients. Spermidine, a polyamine celebrated for its role in cellular autophagy, has faced intense regulatory scrutiny under the Novel Food Regulation (EU) 2015/2283. This analysis dissects the current EFSA safety assessment status, differentiating between naturally derived wheat germ extracts and synthetic variants, while identifying strategic opportunities for export-oriented manufacturers. For buyers looking for Spermidine trihydrochloride bulk options, it is critical to understand that this synthetic form remains under a stricter evaluation pathway compared to botanical extracts.
Traditional Wheat Germ Extract vs. Spermidine Trihydrochloride
From a regulatory risk management perspective, the most common pitfall for exporters is treating all "Spermidine" ingredients as a single entity. The EFSA provides significantly different pathways based on the source and processing method.
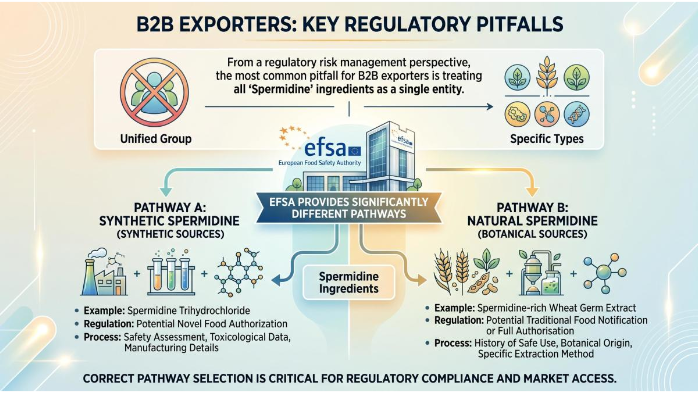
Spermidine-rich Wheat Germ Extract: This is currently the only approved pathway. Certain wheat germ extracts categorized by their polyamine content (specifically spermidine and spermine) have been placed on the Union list of novel foods. The authorization is based on a "traditional" consumption history within the EU, provided the extraction process does not involve concentrations that "significantly alter the nutritional value." For manufacturers seeking Wholesale wheat germ extract spermidine, ensuring that the product stays within the 0.1%–0.5% spermidine concentration range is essential to avoid triggering full Novel Food requirements. Additionally, the rise of Gluten-free wheat germ spermidine variants has opened doors for brands targeting consumers with gluten sensitivities, though gluten removal processes must not alter the polyamine profile.
Synthetic Spermidine (Trihydrochloride): As of late 2023, synthetic spermidine remains under active safety assessment as a "Full" Novel Food application. Unlike botanical extracts, synthetic versions must undergo exhaustive de novo safety testing, including ADME (Absorption, Distribution, Metabolism, and Excretion) studies and genotoxicity screenings. When sourcing Spermidine trihydrochloride bulk, buyers must verify that the supplier has submitted a complete dossier to EFSA; otherwise, selling such products within the EU is prohibited until formal Union List inclusion.
EFSA's Current Safety Appraisal and Scientific Opinions on Spermidine
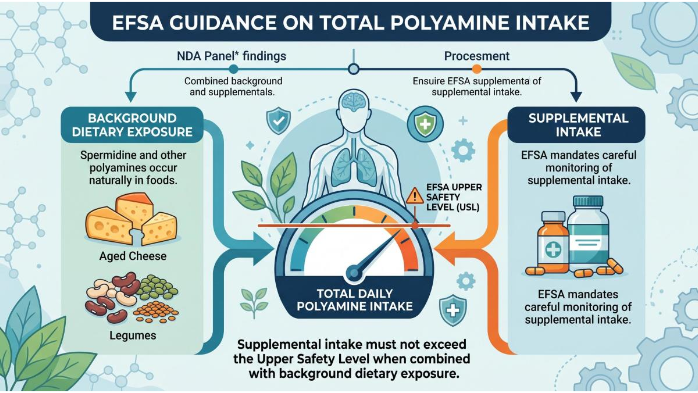
The EFSA Panel on Nutrition, Novel Foods, and Food Allergens (NDA) has expressed specific concerns regarding the total daily intake of polyamines. Because spermidine occurs naturally in foods like aged cheese and legumes, EFSA mandates that supplemental intake does not exceed the "Upper Safety Level" (USL) when combined with background dietary exposure. Interestingly, a Fermented spermidine source—such as those derived from fungal or bacterial fermentation—has recently attracted attention as a potential middle ground. However, EFSA currently classifies any fermented source as a full Novel Food unless the producer can demonstrate a history of safe use within the EU before 1997. Until then, fermented options remain outside the approved pathway.
The Focus on Autophagy and Longevity Claims
While the scientific community is optimistic about spermidine-induced autophagy, EFSA remains highly restrictive on "Health Claims" under Regulation (EC) No 1924/2006. Currently, no authorized health claims exist specifically for spermidine. Brands must use "General Wellness" descriptions to avoid enforcement actions by national authorities. For example, instead of stating "prevents age-related decline," a compliant claim would be "supports normal cellular function." This nuance is especially important for Spermidine bulk powder supplier companies that market directly to European brands; providing compliant claim templates can be a strong value-add.
Traditional Spermidine Extract Compared to Concentrated Polyamines
The following data highlights the regulatory friction based on the concentration of the active ingredient:
Regulatory Risk Gradient:
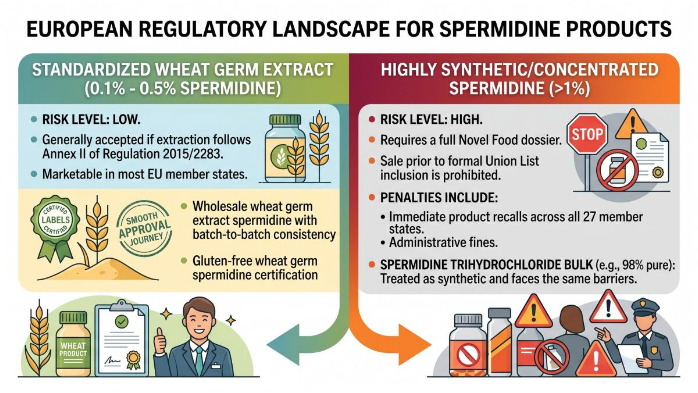
Standardized Wheat Germ Extract (0.1% - 0.5% Spermidine): Low. Generally accepted if extraction follows Annex II of Regulation 2015/2283. Marketable in most EU member states. Here, Wholesale wheat germ extract spermidine with batch-to-batch consistency and Gluten-free wheat germ spermidine certification enjoys a smooth approval journey.
Highly Synthetic/Concentrated Spermidine (>1%):High. Requires a full Novel Food dossier. Sale prior to formal Union List inclusion is prohibited. Penalties include immediate product recalls and administrative fines across all 27 member states. Even Spermidine trihydrochloride bulk at lower purity levels (e.g., 98% pure) is treated as synthetic and therefore faces the same barriers.
Strategic Opportunities for Manufacturers
Manufacturers who can provide "Batch Consistency" data and a "Non-Novel" declaration for their traditional extracts possess a massive competitive advantage. The focus should be on "Primary Material Identification" to prove the ingredient is a food-grade wheat germ byproduct rather than a chemical isolate. A reliable Spermidine bulk powder supplier will differentiate itself by offering full traceability from wheat germ sourcing to final powder, including third-party testing for polyamine levels and gluten content for those requiring Gluten-free wheat germ spermidine. Meanwhile, forward-thinking producers are exploring a Fermented spermidine source as a future-proof investment—though currently not approved, fermentation can offer a vegan, non-wheat alternative once EFSA finalizes its opinion.
The Road to EU Market Leadership
The European market for spermidine is not closed, but it is strictly filtered. The "Wheat Germ" identity is currently the only safe harbor for brands looking to launch immediately. Synthetic entrants, including any Spermidine trihydrochloride bulk products, must wait for the finalized NDA Scientific Opinion, which is expected to clarify the permissible Daily Value (DV) for dietary supplements. For now, smart B2B players should partner with an experienced Spermidine bulk powder supplier that offers Wholesale wheat germ extract spermidine with clear Gluten-free wheat germ spermidine documentation, while keeping an eye on emerging Fermented spermidine source technologies.
Leadingnutra the manufacturer of high-purity Spermidine and specialty nutraceutical ingredients. Contact us for compliant, third-party tested raw materials and expert regulatory guidance.Email: lily@leadingchemical.com | Web: https://www.leadingnutra.com/Spermidine.html
[1]EFSA Summary of Novel Food Applications
[2]Regulation (EU) 2015/2283 on Novel Foods
[3]EIT Food - Spermidine Autophagy Research
[4] EFSA Novel Food Catalog (updated 2024)
[5]Regulation (EC) No 1924/2006 on Nutrition and Health Claims
English
Japanese
Russian
Korean
गोंगेन हें नांव
Deutsch
Corsu
Guarani
Hausa
Cymraeg
Nederlands
Aymara
Français
Kreyòl ayisyen
čeština
ʻŌlelo Hawaiʻi
डोग्रिड ने दी
ภาษาไทย
հայերեն
فارسی
Hmoob
ދިވެހި
भोजपुरी
繁體中文
Türkçe
हिंदी
беларускі
български
tur
Gaeilge
ગુજરાતી
Magyar
Eesti keel
بالعربية
বাংলা
Azərbaycan
Português
Suid-Afrikaanse Dutch taal
کوردی-سۆرانی
Ελληνικά
español
Frysk
dansk
አማርኛ
Bamanankan
euskara
Italiano
Tiếng Việt
অসমীয়া
català
Suomalainen
Eʋegbe
Hrvatski
Cebuano
Gàidhlig na h-Alba
bosanski
galego




