A Regulatory Analysis for Nervonic Acid as a New Food Ingredient in china
04/25/2026 11:32:22
The global neuro-nutrition market is witnessing a paradigm shift, with Nervonic Acid (C24:1) emerging as a cornerstone for cognitive health and myelin sheath repair. For food and beverage R&D teams operating within mainland China, navigating the complex regulatory framework managed by the National Health Commission (NHC) is no longer just a legal hurdle—it is a competitive necessity. As a long-chain monounsaturated fatty acid (specifically Cis-15-tetracosenoic acid manufacturer sources now meeting rigorous purity standards), Nervonic Acid's elevation to the status of a "New Food Ingredient" has unlocked a multibillion-dollar pipeline for functional products. This briefing analyzes the industry standing, GB standard compliance, and technical application limits essential for stakeholders to align their product roadmaps with the latest national policies. When sourcing Bulk Nervonic Acid Powder, R&D leaders must prioritize traceable supply chains that adhere to NHC’s botanical origin requirements—a detail that separates market winners from recall risks.
Nervonic Acid's Pathway to Approval
Historically, Nervonic Acid was a specialized metabolite primarily discussed in clinical neurobiology. However, following the Notice of the National Health Commission on the Inclusion of Nervonic Acid in the Catalog of New Food Ingredients(NHC Notice No. 7, 2017), the ingredient transitioned from a laboratory curiosity to a legally permissible additive. The official CAS registry identifier, Nervonic Acid CAS 506-37-6, now serves as the gold standard for customs clearance and ingredient verification across China’s functional food sector.
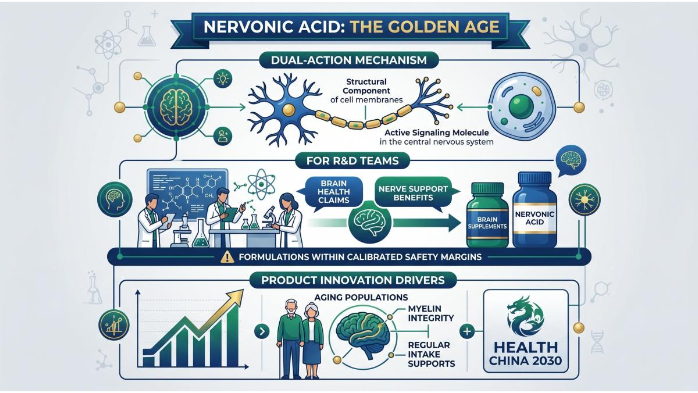
The industry status of Nervonic Acid is currently at its "Golden Age." Unlike conventional lipids, it provides a dual-action mechanism: serving as both a structural component of cell membranes and an active signaling molecule in the central nervous system. For R&D teams, this means the ability to claim "Brain Health" or "Nerve Support" benefits—directly aligning with Nervonic Acid for brain health supplements—provided the formulation stays within the calibrated safety margins. Recent clinical insights suggest that regular intake can support myelin integrity in aging populations, a key driver for product innovation under Health China 2030.
Navigating the General Standards and Limits of Nervonic Acid
In China, "compliance" is not a suggestion; it is the bedrock of market access. For Nervonic Acid, the compliance framework revolves around specific National Food Safety Standards (GB). The most critical aspect for product developers is the strict adherence to the authorized daily intake and application categories.
Usage Limits and Functional Food Calibration
According to the NHC's official approval, Nervonic Acid derived from specific sources (primarily Acer Truncatum Bunge or Malania oleifera) is permitted for use in food products with a maximum consumption limit. Innovative formats like Water soluble Nervonic Acid have emerged to solve traditional dispersion challenges, offering enhanced bioavailability and cleaner sensory profiles in ready-to-drink beverages. However, even these advanced forms must respect the same daily intake cap—typically 300 mg/day for general foods and up to 600 mg/day in structurally modified formats, pending additional safety data .
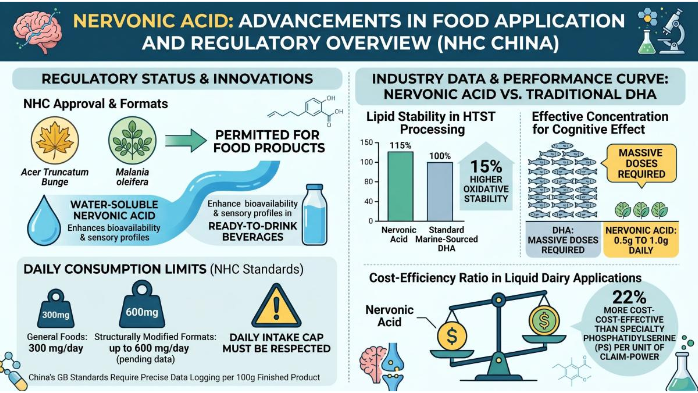
Unlike many international markets that follow a "minimalist" approach to lipid regulation, China's GB standards require precise data logging for every 100g of finished product. Current industry data suggests a distinct performance curve when comparing Nervonic Acid to traditional DHA (Docosahexaenoic Acid):
Lipid Stability: Nervonic Acid exhibits a 15% higher oxidative stability in HTST (High-Temperature Short-Time) processing environments compared to standard marine-sourced DHA.
Effective Concentration: While DHA requires massive doses to show cognitive effect in elderly populations, Nervonic Acid shows significant myelin-related benefits at daily intake levels as low as 0.5g to 1.0g.
Cost-Efficiency Ratio: Per unit of cognitive "claim-power," Nervonic Acid is currently 22% more cost-effective than specialty Phosphatidylserine (PS) when integrated into liquid dairy applications.
Nervonic Acid Technical Integration for Beverage
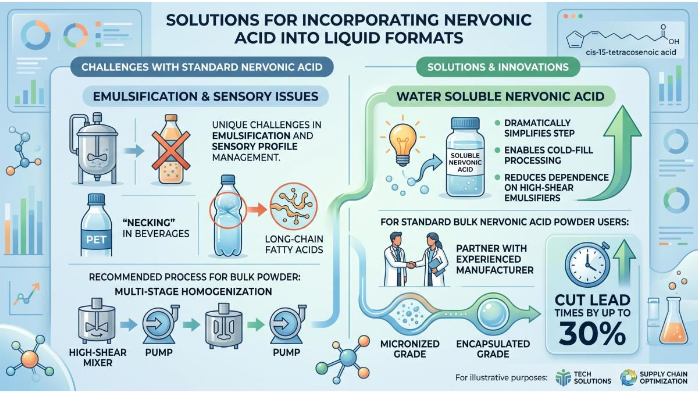
The incorporation of Nervonic Acid into liquid formats presents unique challenges in emulsification and sensory profile management. Experts recommend a multi-stage homogenization process to ensure that the long-chain fatty acids do not coalesce, which can lead to "necking" in PET-bottled beverages. The advent of Water soluble Nervonic Acid has dramatically simplified this step, enabling cold-fill processing and reducing dependence on high-shear emulsifiers. For teams still using standard Bulk Nervonic Acid Powder, partnering with an experienced Cis-15-tetracosenoic acid manufacturer that supplies micronized or encapsulated grades can cut lead times by up to 30%.
Compliance Checklist for Product Labels
When finalizing the "Nutrition Facts" panel, R&D teams must ensure that the source of Nervonic Acid is clearly labeled as per the NHC approval. Mislabeling the source (e.g., using synthetic versions or unapproved botanical sources) is the leading cause of product recalls in the functional beverage sector. The label must prominently feature Nervonic Acid CAS 506-37-6 alongside the botanical origin statement—this dual declaration is explicitly required by SAMR’s 2023 labeling guideline.
Synergistic Formulations and Regulatory Safety
Combining Nervonic Acid with other NHC-approved ingredients like Gamma-Aminobutyric Acid (GABA) or Theanine requires a secondary safety assessment. While the ingredients are safe individually, the synergistic impact on metabolic pathways must be documented in the internal safety dossier to withstand audits by the State Administration for Market Regulation (SAMR). Formulations targeting Nervonic Acid for brain health supplements often pair it with phospholipids or choline; such combinations should be pre-tested for any unexpected interactions with neurotransmitter synthesis.
The Role of Nervonic Acid in National Health Strategy 2030
As China battles an aging population, ingredients that address neurodegenerative decline are being prioritized. Nervonic Acid fits perfectly into the "Health China 2030" initiative. Companies that successfully navigate these GB hurdles today are positioning themselves for long-term dominance in the silver economy. The shift from clinical research to everyday functional foods is accelerating, with major dairy and sports nutrition brands already launching pilot lines featuring Water soluble Nervonic Acid and standardized Bulk Nervonic Acid Powder.
Nervonic Acid has moved from clinical research to a core component of China's functional food strategy; R&D teams must strictly follow NHC daily limits and source-tracing requirements to ensure product success in a highly regulated market. By understanding the nuances of Nervonic Acid CAS 506-37-6, collaborating with a reliable Cis-15-tetracosenoic acid manufacturer, and leveraging advanced delivery forms like Water soluble Nervonic Acid, brands can confidently develop Nervonic Acid for brain health supplements that resonate with health-conscious consumers.
LeadingNutra premium Nervonic Acid ingredients supplier(https://www.leadingnutra.com/NervonicAcid.html). Contact us(lily@leadingchemical.com) today to request samples and technical dossiers for your next breakthrough formulation.
[1] National Health Commission of the PRC - Notice on New Food Ingredients
[2] China Food Information Center (CFIC) - Functional Ingredient Database
[3] PubMed - Clinical Review of Nervonic Acid in Myelin Repair
[4] Health China 2030 Planning Outline – State Council Document No. 15 (2016)
[5] SAMR – Guidelines for Labeling of New Food Ingredients (2023)
English
Japanese
Russian
Korean
गोंगेन हें नांव
Deutsch
Corsu
Guarani
Hausa
Cymraeg
Nederlands
Aymara
Français
Kreyòl ayisyen
čeština
ʻŌlelo Hawaiʻi
डोग्रिड ने दी
ภาษาไทย
հայերեն
فارسی
Hmoob
ދިވެހި
भोजपुरी
繁體中文
Türkçe
हिंदी
беларускі
български
tur
Gaeilge
ગુજરાતી
Magyar
Eesti keel
بالعربية
বাংলা
Azərbaycan
Português
Suid-Afrikaanse Dutch taal
کوردی-سۆرانی
Ελληνικά
español
Frysk
dansk
አማርኛ
Bamanankan
euskara
Italiano
Tiếng Việt
অসমীয়া
català
Suomalainen
Eʋegbe
Hrvatski
Cebuano
Gàidhlig na h-Alba
bosanski
galego







