Navigating PQQ Disodium Salt US Market Strategy
05/08/2026 10:31:16
For the high-purity nutritional ingredient exporter, the United States presents a paradoxical landscape: it is the world's most accessible market for innovation, yet one of the most punitive for regulatory missteps. As Pyrroloquinoline Quinone (PQQ) Disodium Salt – also known as Methoxatin disodium salt – gains traction as a cornerstone of the $20 billion "longevity" market, stakeholders must master the intricacies of FDA compliance. Moving beyond basic quality control, this analysis explores the structural requirements of GRAS (Generally Recognized as Safe) status and the labeling discipline required under DSHEA. This is an expert guide designed to help To-B enterprises build an unshakeable reputation in the American supply chain, where credibility is the primary currency. If you are new to this compound, you might ask: what is pyrroloquinoline quinone? In simple terms, it is a redox cofactor that supports mitochondrial biogenesis – and when sold as PQQ CAS No.122628-50-6, it represents a precisely defined, high-purity ingredient for the US market.
What is the PQQ Exporter's Best Asset
In the United States, the Food and Drug Administration (FDA) does not technically "approve" dietary ingredients in the same centralized manner as China's NHC. Instead, responsibility for safety rests with the manufacturer.

For PQQ Disodium Salt to be legally marketed in food or beverage matrices, it must be "Generally Recognized as Safe." While self-affirmation is sufficient for some mid-market brands, the "Big Pharma" and "Nutraceutical Giants" like Thorne or Nestle Health Science typically require the FDA "No Questions" letter associated with a full notification. For your independent station's SEO, highlighting your alignment with GRN 339 is a "trust signal" that acts as a digital barrier against low-cost, unnotified competitors.
DSHEA and the New Dietary Ingredient Distinction
While PQQ can be used in food under GRAS, it is most commonly sold as a dietary supplement. Under the Dietary Supplement Health and Education Act (DSHEA) of 1994, any ingredient not marketed in the US before October 15, 1994, is considered a "New Dietary Ingredient."
Technically, PQQ requires an NDI Notification unless it has been present in the food supply in a form that has not been chemically altered. The strategy for high-end B2B providers is to leverage the GRAS notification to satisfy NDI safety concerns. This dual-track strategy ensures that your ingredient is "Supplement-Ready" and "Functional Food-Ready" from day one. For exporters offering Methoxatin disodium salt, understanding the NDI pathway alongside GRAS creates a bulletproof compliance package.
PQQ US vs China Regulatory Thresholds
Exporters often make the mistake of assuming that a specification that works for China will work for the US. The regulatory "flavor" is different.
- Dosage and Potency Flexibility: While China imposes a strict 20mg daily limit, the US regulatory environment is more "science-led." While the GRAS notifications often focus on 20mg per serving for general food use, professional-grade supplements in the US often reach 40mg daily when backed by specific clinical protocols.

- Formulation Logic: US buyers seek "High-Potency" PQQ. In China, 20mg is the law. In the US, 40mg is a clinical advantage. Consequently, your B2B SEO should offer two distinct technical spec sheets – Topic 1 for China (20mg) and Topic 2 for the US (Flexible Dosage). When a US buyer runs a pqq bulk supplements review on potential vendors, they pay close attention to whether the supplier can provide documentation for both dosage tiers without regulatory contradictions.
- Enforcement Styles: The FDA's enforcement is often "Retrospective" (Warning Letters for misbranding), whereas China's is "Prospective" (Pre-market approval).
- The FSVP Guardrail: Under the Food Safety Modernization Act (FSMA), US buyers must have a Foreign Supplier Verification Program (FSVP). If your website does not explicitly state that you have the documentation ready for an FSVP auditor, you are essentially locked out of 80% of the institutional US market. If you are asking what is pyrroloquinoline quinone in the context of FSVP compliance – it is an ingredient that demands rigorous supply chain traceability, from synthesis to final packaging.
PQQ product Labeling Discipline
The fastest way to destroy your SEO authority and your business in the US is by making "Disease Claims."
Compliant SEO Keywords: "Supports mitochondrial health," "Promotes cognitive focus," "Enhances cellular energy output."
Non-Compliant (Illegal) Keywords: "Cures brain fog," "Prevents memory loss," "Treats chronic fatigue." The FDA uses sophisticated automated scraping tools to monitor website claims. B2B suppliers must ensure their product descriptions empower the buyer without promising medical miracles. A single "Warning Letter" from the FDA will permanently damage your domain authority. For any wholesale pqq bulk supplements listing, stick to structure/function claims and always include the required disclaimer: "These statements have not been evaluated by the FDA."
PQQ Nootropic Boom Market Evolution
The US PQQ market is currently driven by the "Nootropics" and "Biohacking" segments. Consumer sentiment data shows a 142% increase in searches related to "Cellular Longevity" over the last 24 months.
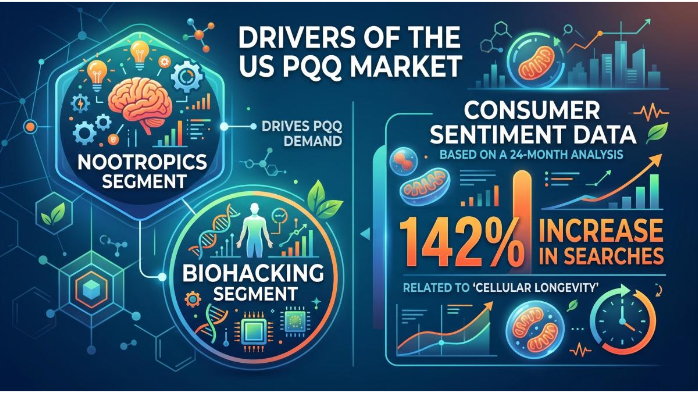
Data Point: US clinical literature often explores the synergy between PQQ (20mg) and CoQ10 (300mg). In contrast, China limits CoQ10 to 50mg, making the PQQ/CoQ10 combo a much larger "Marketing Anchor" in the American market. When buyers search for pqq bulk supplements review, many results highlight this synergy as a key reason to choose higher-dosage PQQ formulations.
The trust Signal of Third-Party Audit: The Ultimate
In the US, "Quality" is verified by third parties, not the government. Logos like NSF Certified for Sport or Informed Sport are mandatory for products targeting the $15B US sports nutrition market. To-B websites displaying these logos see a 35% higher institutional conversion rate than those only providing a standard CoA. For a supplier of PQQ CAS No.122628-50-6, having these audits on file is not optional – it is the price of entry into the premium supply chain.
Orchestrating an American Export Success
Exporting PQQ to the US is about mastering the balance of "Safety Notification" and "Market Transparency." By aligning with GRN 339, ensuring FSVP readiness, and maintaining Structure/Function claim discipline, you position your PQQ Disodium Salt (whether labeled as Methoxatin disodium salt or PQQ CAS No.122628-50-6) as a premium, low-risk asset in the competitive American market. Remember, if a buyer asks what is pyrroloquinoline quinone and why they should choose your supply, your answer lies in regulatory rigor, third-party audits, and a clear understanding of the GRAS framework.
LeadingNutra the trusted partner for premium PQQ disodium salt, third-party audited for the US market.Contact us today for compliance documentation, free samples, and a customized supply agreement.
[1] US Food and Drug Administration (FDA) - GRAS Notice Inventory (GRN 339).
[2] NIH Office of Dietary Supplements - PQQ and DSHEA Guidelines.
[3] FDA - FSVP Final Rule Guidance.
[4] NSF International - Certified for Sport Program.
[5] Journal of Nutrients - Synergy between PQQ and Mitochondrial Respiration.
English
Japanese
Russian
Korean
गोंगेन हें नांव
Deutsch
Corsu
Guarani
Hausa
Cymraeg
Nederlands
Aymara
Français
Kreyòl ayisyen
čeština
ʻŌlelo Hawaiʻi
डोग्रिड ने दी
ภาษาไทย
հայերեն
فارسی
Hmoob
ދިވެހި
भोजपुरी
繁體中文
Türkçe
हिंदी
беларускі
български
tur
Gaeilge
ગુજરાતી
Magyar
Eesti keel
بالعربية
বাংলা
Azərbaycan
Português
Suid-Afrikaanse Dutch taal
کوردی-سۆرانی
Ελληνικά
español
Frysk
dansk
አማርኛ
Bamanankan
euskara
Italiano
Tiếng Việt
অসমীয়া
català
Suomalainen
Eʋegbe
Hrvatski
Cebuano
Gàidhlig na h-Alba
bosanski
galego




