EU Novel Food Compliance for Urolithin A Wholesale
05/02/2026 16:10:03
Entering the European Union market with advanced dietary supplements requires navigating a regulatory environment characterized by extreme rigor and uncompromising safety standards. For suppliers and brand manufacturers dealing in high-tech functional ingredients, the European Food Safety Authority (EFSA) represents both a formidable barrier to entry and a mark of ultimate clinical validation. Urolithin A, a gut microbiome-derived metabolite of ellagitannins recognized for its profound impact on mitochondrial health and mitophagy, has rapidly shifted from a niche anti-aging compound to a highly sought-after commercial ingredient. Whether you are looking for Bulk Urolithin A Powder for large-scale formulation or a reliable Urolithin A Raw Material Supplier, understanding EFSA’s requirements is the first step toward success. However, because this specific compound lacks a significant history of human consumption within the European Union prior to May 15, 1997, it falls strictly under the jurisdiction of the Novel Food Regulation (EU) 2015/2283. Securing market authorization requires comprehensive toxicological profiling, precise manufacturing controls, and a flawless regulatory dossier. This guide breaks down the compliance pathway for synthetic Urolithin A, focusing on EFSA's safety assessment protocols, toxicological thresholds, and the strategic steps necessary for B2B enterprises to achieve regulatory approval and commercialize this high-value ingredient in the stringent European market.
Strategic EU Novel Food Context for Urolithin A Wholesale
The European Union operates on a pre-market authorization system for new food ingredients. Unlike the United States, where the Generally Recognized As Safe (GRAS) self-affirmation pathway allows companies to bring ingredients to market relatively quickly, the EU Novel Food framework mandates a centralized, exhaustive evaluation by EFSA before any commercial activity can commence. For a high-tech ingredient like Urolithin A, this means every aspect of its production, composition, and biological interaction must be documented and defended. B2B players engaged in Urolithin A Wholesale must recognize that the EU approval not only legalizes the ingredient but also builds trust with premium buyers who demand full traceability and safety.
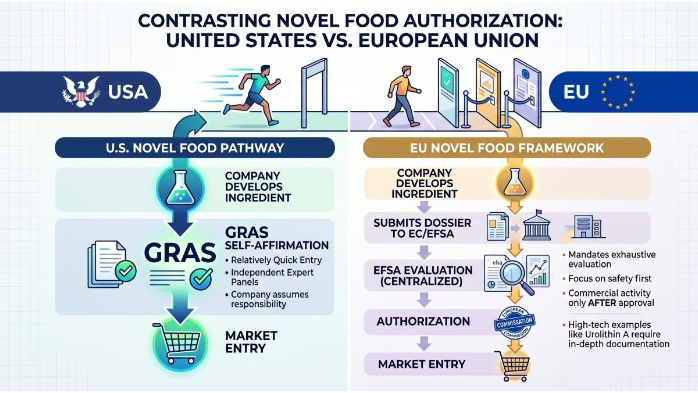
The commercial rationale for pursuing this demanding pathway is substantial. A positive EFSA opinion and subsequent authorization by the European Commission grant an ingredient legal status across all member states, providing access to a massive demographic of health-conscious, aging consumers. Furthermore, under Article 26 of the Novel Food Regulation, applicants can request a five-year period of data protection. If granted, this exclusivity prevents generic competitors from leveraging the original applicant's proprietary toxicological and clinical data to secure their own approvals, offering a critical competitive moat for pioneering suppliers. Smart Urolithin A Ingredient Sourcing strategies therefore prioritize suppliers with existing or in-progress EFSA dossiers.
EFSA Safety Assessment Core for Urolithin A Compliance
When evaluating synthetic Urolithin A, EFSA's Panel on Nutrition, Novel Foods and Food Allergens (NDA) focuses primarily on ensuring that the ingredient does not pose a safety risk to human health at the proposed intake levels. Because the ingredient is manufactured via chemical synthesis rather than extracted from botanical sources like pomegranates, the assessment heavily scrutinizes the purity of the final product and the potential presence of manufacturing byproducts.
Compositional Data and Manufacturing Rigor
EFSA requires exhaustive compositional data demonstrating that the manufacturing process is highly controlled and capable of producing a consistent product. Suppliers must submit analytical data from at least five independent, representative batches of the synthetic Urolithin A. Some innovative manufacturers have developed a Water-soluble Urolithin A version to enhance bioavailability and ease of formulation; however, any modified form requires its own safety assessment or an extension of use, as EFSA’s current authorization applies strictly to the synthetic, non-modified compound.
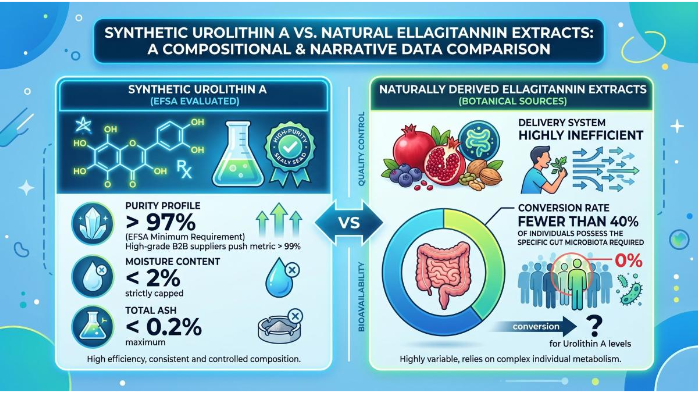
When analyzing the compositional requirements, a stark narrative data comparison emerges between synthetic Urolithin A and naturally derived ellagitannin extracts. The synthetic Urolithin A evaluated by EFSA is required to achieve a purity profile exceeding 97 percent, with many high-grade suppliers pushing this metric above 99 percent. Moisture content is strictly capped at less than 2 percent, and total ash must remain below 0.2 percent. In contrast, attempting to rely on botanical extracts yields a highly inefficient delivery system; clinical data indicates that fewer than 40 percent of individuals possess the specific gut microbiota required to convert natural ellagitannins into meaningful levels of Urolithin A. By utilizing the highly purified synthetic form, manufacturers bypass the variable human microbiome entirely, delivering a precise, standardized dosage. Furthermore, EFSA imposes strict limits on environmental and manufacturing contaminants. For instance, heavy metal thresholds are rigorously defined: lead must not exceed 0.5 milligrams per kilogram, arsenic is capped at 1 milligram per kilogram, and cadmium and mercury are restricted to 0.5 and 0.1 milligrams per kilogram, respectively. Solvent residues, such as dimethyl sulfoxide or acetic acid used during synthesis, must also fall well below established toxicological thresholds of concern. For any Urolithin A Raw Material Supplier, meeting these compositional limits is non-negotiable for market access.
Toxicological Profiling and Margin of Safety
The cornerstone of the EFSA Novel Food dossier is the toxicological safety data. For synthetic Urolithin A, the applicant must provide a comprehensive battery of tests, typically conducted in compliance with Good Laboratory Practice (GLP) standards. This includes in vitro genotoxicity testing—such as the bacterial reverse mutation assay (Ames test) and the in vitro mammalian chromosomal aberration test—to unequivocally prove that the compound does not damage DNA.
Following genotoxicity clearance, EFSA relies heavily on subchronic toxicity studies to establish a safe intake level for humans. In a pivotal 90-day oral toxicity study conducted on rats, researchers administered varying doses of synthetic Urolithin A to observe potential adverse physiological, biochemical, or histological changes. The narrative data comparison between the animal testing thresholds and the proposed human dosage reveals a highly favorable safety profile. During the 90-day study, the highest dose tested—which resulted in no adverse effects—was established as the No Observed Adverse Effect Level (NOAEL). This NOAEL was determined to be 3,451 milligrams per kilogram of body weight per day for male rats and 3,826 milligrams per kilogram of body weight per day for female rats. When EFSA juxtaposed this animal NOAEL against the proposed maximum human intake of 500 milligrams per day (which equates to roughly 7.1 milligrams per kilogram of body weight per day for a standard 70-kilogram adult), the resulting margin of exposure was calculated to be nearly 500-fold. In toxicological assessments, a margin of exposure exceeding 100 is generally considered indicative of a robust safety buffer, meaning the 500-fold margin for Urolithin A provided the NDA Panel with exceptional confidence in its safety for human consumption. Companies offering Bulk Urolithin A Powder must ensure that every batch replicates the purity and safety profile used in these toxicological studies.
Absorption, Distribution, Metabolism, and Excretion (ADME)
To further satisfy EFSA's requirements, applicants must detail the ADME profile of Urolithin A. Clinical and preclinical data demonstrate that following oral administration, Urolithin A is rapidly absorbed in the gastrointestinal tract and subsequently undergoes extensive phase II metabolism in the liver, primarily via glucuronidation and sulfation. The predominant circulating forms in the human bloodstream are Urolithin A glucuronide and Urolithin A sulfate. EFSA's evaluation of these metabolic pathways confirms that the compound does not accumulate toxically in mammalian tissues and is efficiently excreted, primarily through urine, further supporting its safety for long-term daily supplementation. A Water-soluble Urolithin A derivative would alter these ADME properties, which is why EFSA requires separate data for any such variant.
GRAS Urolithin A Target Population and Use Conditions
Regulatory compliance is not solely about proving a substance is safe; it is equally about defining exactly who will consume it and in what quantities. Based on the submitted toxicological and clinical data, EFSA concluded that synthetic Urolithin A is safe for the general adult population. The authorized conditions of use permit its inclusion in standard food supplements at a maximum dose of 500 milligrams per day. Additionally, it is authorized for use in foods for special medical purposes (FSMP) at the same maximum daily dosage.
However, EFSA mandates specific labeling requirements to protect vulnerable demographics. Because the safety dossier lacked specific developmental and reproductive toxicity studies encompassing pregnancy and lactation, the Novel Food authorization explicitly excludes pregnant women, lactating women, and children under the age of 18. Suppliers and consumer-facing brands must ensure that product labels clearly state these contraindications to remain compliant with EU law. For efficient Urolithin A Ingredient Sourcing, it is vital to work with partners who provide clear documentation on target populations and usage limits.
Regulatory Pathway Guide for Urolithin A Powder Supplier
For suppliers aiming to introduce their own synthesized Urolithin A to the European market, understanding the administrative timeline and procedural nuances is critical. If a supplier's manufacturing process or final specification differs significantly from the already authorized Urolithin A, they cannot simply piggyback on the existing Novel Food status. They must submit their own dossier or apply for an extension of use. This is particularly relevant for those developing Water-soluble Urolithin A or other novel formats.
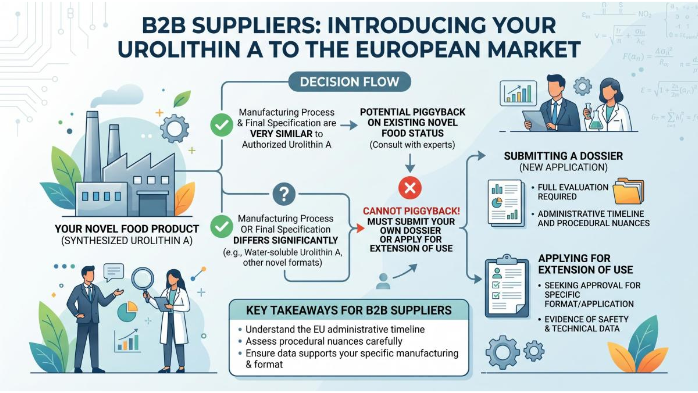
Dossier Preparation and Submission
The preparation of a Novel Food dossier is a resource-intensive endeavor that requires multidisciplinary expertise. Suppliers must compile detailed technical dossiers that include full chemical characterization, descriptions of the production process, stability data (typically demonstrating shelf-life under both standard and accelerated conditions), and comprehensive toxicological and human clinical data. The stability data must prove that Urolithin A does not degrade into unknown or harmful byproducts over a proposed shelf life of 24 to 36 months. A trustworthy Urolithin A Raw Material Supplier will have these dossiers ready or under development, saving downstream buyers years of regulatory work.
Navigating the Administrative Timeline
The EU Novel Food approval process is notoriously lengthy. Once a dossier is submitted via the European Commission's e-submission portal, it undergoes a suitability and validation check, which can take several months. If validated, the dossier is forwarded to EFSA for the scientific risk assessment phase. By law, EFSA has nine months to issue its opinion, but this clock stops whenever EFSA requests supplementary information from the applicant. It is highly common for the NDA Panel to halt the clock to request clarification on batch analyses or toxicological methodologies.
Upon the publication of a positive EFSA opinion, the process transitions to the risk management phase. The European Commission has seven months to draft an implementing regulation, present it to the Standing Committee on Plants, Animals, Food and Feed (PAFF), and officially add the ingredient to the Union List of Novel Foods. For suppliers, this means the entire timeline from dossier submission to market authorization typically spans 18 to 24 months, assuming the initial data package is exceptionally robust. Engaging in Urolithin A Wholesale requires planning ahead—buyers should start sourcing from applicants who have already secured approval or are well-advanced in the process.
Bridging Compliance to Market Entry for Urolithin A Wholesale
Achieving EFSA approval for Urolithin A is a monumental milestone, but it is only the first step in successful market entry. Suppliers must transition from regulatory compliance to commercial scale-up without compromising the strict parameters defined in their Novel Food dossier. This requires the implementation of state-of-the-art Good Manufacturing Practices (GMP) and continuous quality control monitoring.
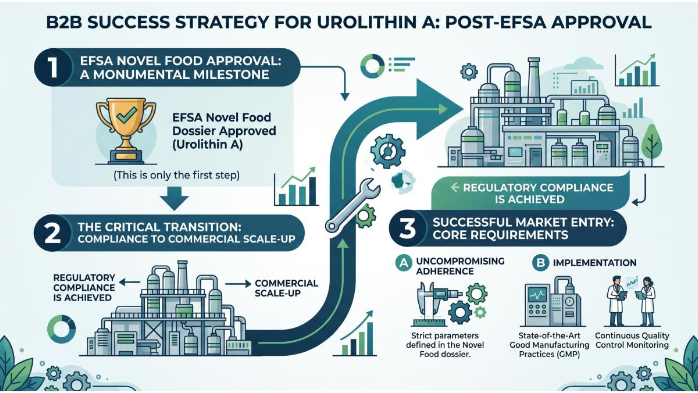
European buyers—ranging from specialized nutraceutical brands to large-scale pharmaceutical entities—conduct rigorous due diligence before onboarding a new ingredient supplier. They will demand comprehensive Certificates of Analysis (CoA) that perfectly align with the EFSA-approved specifications. Any deviation in purity, heavy metal content, or solvent residues can result in rejected shipments and severe regulatory penalties. Therefore, Suppliers must treat the EFSA specifications not merely as a regulatory hurdle, but as the foundational blueprint for their commercial manufacturing operations. For those involved in Urolithin A Ingredient Sourcing, choosing a supplier that can consistently deliver Bulk Urolithin A Powder meeting these specifications is the key to long-term success.
The stringent nature of the EU market serves as a global benchmark. A synthetic Urolithin A ingredient that successfully navigates the EFSA Novel Food pathway carries an implicit seal of safety and quality that resonates far beyond Europe's borders. It facilitates easier market entry in other jurisdictions and commands a premium in the competitive landscape of healthy aging and cellular nutrition. By meticulously aligning manufacturing processes with EFSA's rigorous toxicological and compositional demands, Suppliers can unlock the vast commercial potential of Urolithin A and establish themselves as leaders in the high-tech dietary supplement industry.
Leadingnutra premium partner for EFSA-compliant Urolithin A(https://www.leadingnutra.com/UrolithinA.html) raw materials. Contact us today for certified bulk powders, technical support, and hassle-free wholesale solutions.
[1] European Food Safety Authority (EFSA). "Safety of synthetic urolithin A as a novel food pursuant to Regulation (EU) 2015/2283." EFSA Journal.
[2] U.S. Food and Drug Administration (FDA). "GRAS Notice (GRN) No. 791: Urolithin A."
[3] Andreux, P. A., et al. "The mitophagy activator urolithin A is safe and induces a molecular signature of improved mitochondrial and cellular health in humans." Nature Metabolism 1, 595–603 (2019).
[4] European Commission. "Union List of Novel Foods – Urolithin A entry."
[5] D'Amico, D., et al. "Impact of Urolithin A on muscle strength and mitochondrial biomarkers in elderly adults: A double-blind randomized controlled trial." Cell Reports Medicine, 2022.
[6] EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA). "Scientific opinion on the safety of synthetic urolithin A – supplementary data on genotoxicity." EFSA Journal 2021;19(12):6933.
English
Japanese
Russian
Korean
गोंगेन हें नांव
Deutsch
Corsu
Guarani
Hausa
Cymraeg
Nederlands
Aymara
Français
Kreyòl ayisyen
čeština
ʻŌlelo Hawaiʻi
डोग्रिड ने दी
ภาษาไทย
հայերեն
فارسی
Hmoob
ދިވެހި
भोजपुरी
繁體中文
Türkçe
हिंदी
беларускі
български
tur
Gaeilge
ગુજરાતી
Magyar
Eesti keel
بالعربية
বাংলা
Azərbaycan
Português
Suid-Afrikaanse Dutch taal
کوردی-سۆرانی
Ελληνικά
español
Frysk
dansk
አማርኛ
Bamanankan
euskara
Italiano
Tiếng Việt
অসমীয়া
català
Suomalainen
Eʋegbe
Hrvatski
Cebuano
Gàidhlig na h-Alba
bosanski
galego





