EU Novel Food Compliance Guide for Bulk L-theanine Powder
04/29/2026 09:35:03
For international suppliers and European distributors of nutritional supplements, the regulatory status of L-Theanine within the European Union represents one of the most critical hurdles for market entry. As a compound marketed as a “Novel Food,” L-Theanine is subject to rigorous safety assessments and specific authorization procedures under Regulation (EU) 2015/2283. Failing to navigate these waters can lead to immediate product recalls, border rejections, and long-term legal repercussions. This is especially true when dealing with Bulk L-theanine powder, which must meet strict purity and documentation standards to avoid seizure at EU borders.
This guide serves as a technical manual for industry stakeholders aiming to align their L-Theanine products with European Food Safety Authority (EFSA) standards. Whether you supply Wholesale L-theanine or specialty isolates, understanding the subtle differences between synthetic and natural extractions is key. We will dissect the nuances between synthetic and natural extractions, examine the pivotal safety dossiers, and provide a roadmap for achieving compliance in a fragmented but lucrative European market.
Understanding Novel Food Bulk L-theanine Powder
In the European regulatory framework, a “Novel Food” is defined as any food that was not consumed to a significant degree by humans in the EU before May 15, 1997. L-Theanine, despite its thousands of years of consumption through green tea, falls under this classification when it is concentrated, purified, or synthetically produced. The logic applied by the European Commission is that the exposure levels of pure L-Theanine isolates (200mg-400mg per capsule) are vastly different from the micro-doses encountered in traditional tea drinking (approx 20-30mg per cup). For businesses offering L-theanine raw material bulk, this classification means you cannot simply export without a validated Novel Food application.
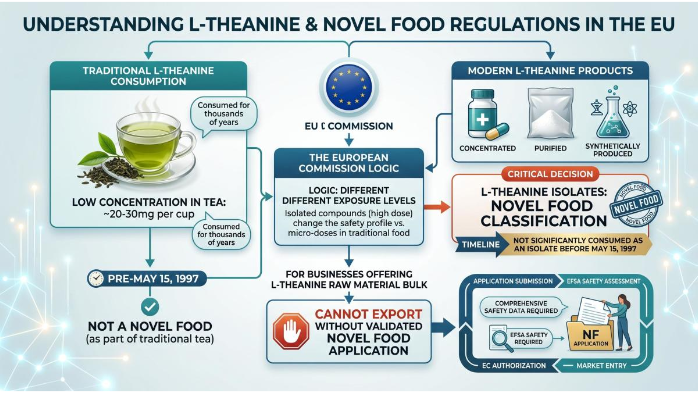
The L-Theanine Natural vs. Synthetic Source
One of the primary friction points in EU compliance is the origin of the L-Theanine.
Natural Extraction: Typically derived from tea leaves through aqueous extraction and chromatographic purification. While “natural,” these products still require verification of a history of use if the final ratio of the compound to other tea components is significantly altered. A popular choice among European buyers is Organic L-theanine tea extract, which not only appeals to clean-label trends but also may face slightly reduced scrutiny on residual solvents compared to synthetic versions.
Synthetic L-Theanine: Produced via organic synthesis (e.g., using ethylamine and glutamine) or enzymatic reaction. This is strictly categorized as a Novel Food and cannot be marketed until the specific manufacturer receives a Union List authorization. Even then, the authorization is often manufacturer-specific for the first 5 years under data protection rules. For those handling Non-GMO L-theanine bulk, it is critical to note that “non-GMO” does not exempt you from Novel Food requirements – EFSA evaluates safety regardless of genetic modification status.
Navigating EFSA Safety for Bulk L-theanine Powder
The EFSA (European Food Safety Authority) is the gatekeeper for Novel Food approval. Their assessments focus on toxicological data, stability, production process, and intended use levels. To successfully register Bulk L-theanine powder, suppliers must submit complete technical dossiers that include stability studies under EU climate conditions.
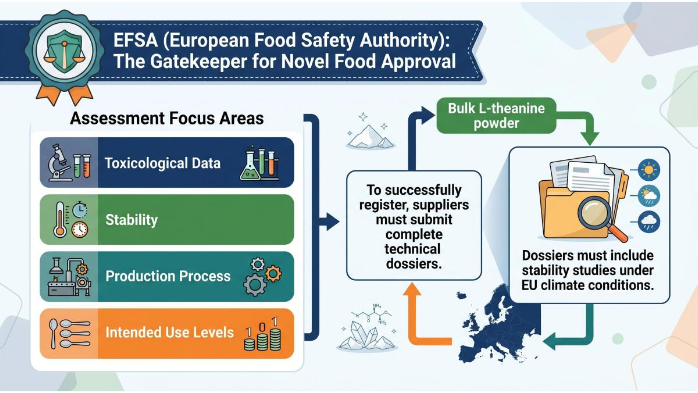
Key Safety Dossiers and Recent Rulings
In recent years, the EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA) has ramped up its scrutiny. Dossiers must include ADME studies (Absorption, Distribution, Metabolism, and Excretion).A recent evaluation of synthetic L-Theanine concluded that safety was established for standard intake in adults, but flagged concerns for “vulnerable populations” such as pregnant women or young children without further data. When you source Wholesale L-theanine from Asian manufacturers, make sure their dossiers address these population-specific gaps; otherwise, your application will be rejected.
Data Comparison: Extraction vs Synthesis Safety Profiles
| Parameter |
Natural Extract |
Synthetic Variant |
| Chemical Purity |
90-98% (depends on residual tea solids) |
99.5%+ |
| Contaminant Risk Profile |
Pesticides, heavy metals (Pb/As), PAHs |
Residual solvents |
| EFSA Scrutiny Level |
Moderate – occasional notification route |
Full technical dossier per Regulation (EU) 2017/2469 + genotoxicity studies |
| Time to Market |
1-2 years (if using existing tea extract framework) |
3-5 years for full authorization |
For companies dealing in L-theanine raw material bulk, natural extracts are often the faster path to market, provided you can prove the extraction process does not introduce novel contaminants. Meanwhile, Organic L-theanine tea extract has the added advantage of aligning with EFSA’s preference for minimal processing residues.
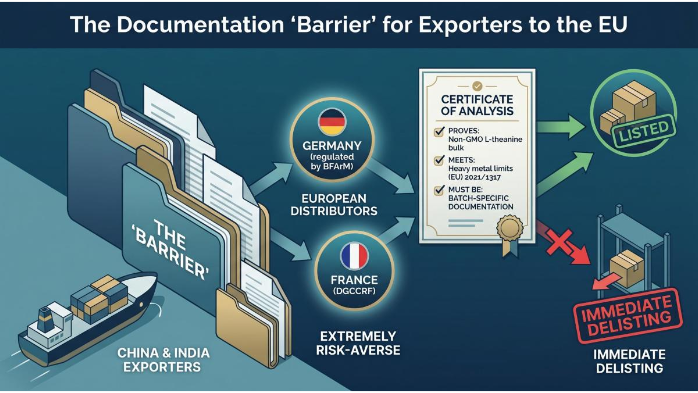
Barriers to European Market for Bulk L-theanine Powder
The “Barrier” for most Chinese and Indian exporters is not just the quality of the product, but the quality of the paper-trail. European distributors, particularly in Germany (regulated by BFArM) or France (DGCCRF), are extremely risk-averse. They will request certificates of analysis that prove your Non-GMO L-theanine bulk meets the heavy metal limits set by Regulation (EU) 2021/1317. Failure to provide batch-specific documentation often results in immediate delisting.
The UK’s Divergent Path (FSA vs EFSA)
Post-Brexit, the UK’s Food Standards Agency (FSA) has its own Novel Food register. Currently, L-Theanine is in a “transition period” in the UK. Any product on the market before 2020 may currently stay while their application is pending. However, in the EU-27, a lack of Union List status means your product – whether it’s Bulk L-theanine powder or finished capsules – can be seized at major ports like Rotterdam or Hamburg under the "General Food Law" . Therefore, if you supply Wholesale L-theanine to both markets, you need dual compliance strategies.
Labeling and Health Claims Restrictions
Even if authorized as a food ingredient, health claims are a separate battle. Under Regulation (EC) No 1924/2006, claims like “L-Theanine reduces stress” or “Natural relaxation” have NOT been approved. Compliant brands must use “Article 10.3” claims, such as “Contains L-Theanine, which supports mental focus,” provided they also include a general allowed claim about a vitamin (like B12 for psychological function) included in the mix. When marketing Organic L-theanine tea extract, you are allowed to use the EU Organic logo (if certified) but still cannot make unauthorized health claims.
Strategic Compliance Roadmap for Bulk L-theanine Powder
To successfully enter the EU market with L-Theanine, B2B exporters should follow these steps:
- Source Natural Derivatives: Prioritize high-purity natural extracts – especially Organic L-theanine tea extract – as they are more “friendly” to European consumer perceptions and regulatory frameworks compared to synthetic lab-made versions. This also simplifies the Novel File.
- Invest in Technical Dossiers: Ensure your COA (Certificate of Analysis) matches the specifications in Regulation (EU) No 231/2012 for food additives where applicable, and exceeds “standard” requirements for heavy metals. For Non-GMO L-theanine bulk, include third-party verification of non-GMO status alongside EFSA-required toxicology data.
- Appoint an EU Responsible Person (RP): Having a legal entity in the EU to handle safety notifications is mandatory for long-term successful trade. This RP will also manage any post-market monitoring for your Bulk L-theanine powder shipments.
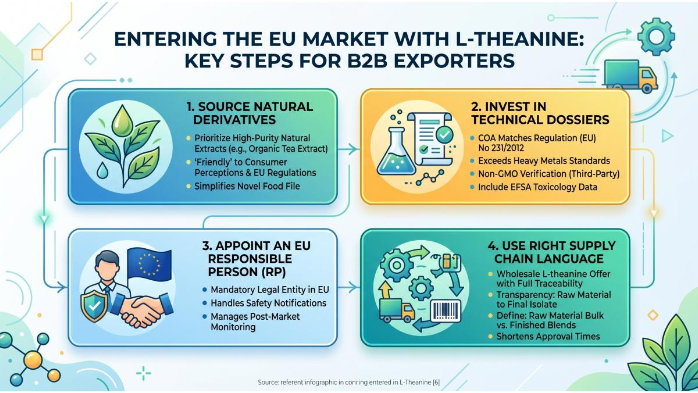
Use the Right Supply Chain Language: When negotiating with European buyers, clearly label your offers as Wholesale L-theanine with full traceability from raw material to final isolate. Transparency on whether you supply L-theanine raw material bulk or finished blends can shorten approval times significantly.
The Cost of Compliance vs. Market Opportunity
The European stress-relief market is projected to reach billions per annum by 2030. While the Novel Food status of L-Theanine presents a significant barrier, it also acts as a “moat” that protects compliant, high-quality brands from low-tier competition. Understanding and adhering to EFSA guidelines is not just about staying legal; it’s about building a premium, sustainable brand in one of the world’s most demanding and profitable markets. Whether you offer Organic L-theanine tea extract, Non-GMO L-theanine bulk, or standard Bulk L-theanine powder, the key is a meticulously prepared dossier and a trusted EU partner.
Leadingnutra the trusted L-Theanine manufacturer(https://www.leadingnutra.com/L-Theanine.html) and regulatory partner for premium ingredients. Contact us today for compliant sourcing solutions and expert support.
[1]EFSA Journal: Safety of L-theanine as a Novel Food.
[2]European Commission: Novel Food Catalogue Entry for L-Theanine.
[3]EUR-Lex: Regulation (EU) 2015/2283 on Novel Foods.
[4]EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA) – Guidance on novel food applications.
[5]European Commission – Novel Food status database (updated 2024).
[6]EU Regulation (EU) 2021/1317 – Maximum levels for contaminants in food.
English
Japanese
Russian
Korean
गोंगेन हें नांव
Deutsch
Corsu
Guarani
Hausa
Cymraeg
Nederlands
Aymara
Français
Kreyòl ayisyen
čeština
ʻŌlelo Hawaiʻi
डोग्रिड ने दी
ภาษาไทย
հայերեն
فارسی
Hmoob
ދިވެހި
भोजपुरी
繁體中文
Türkçe
हिंदी
беларускі
български
tur
Gaeilge
ગુજરાતી
Magyar
Eesti keel
بالعربية
বাংলা
Azərbaycan
Português
Suid-Afrikaanse Dutch taal
کوردی-سۆرانی
Ελληνικά
español
Frysk
dansk
አማርኛ
Bamanankan
euskara
Italiano
Tiếng Việt
অসমীয়া
català
Suomalainen
Eʋegbe
Hrvatski
Cebuano
Gàidhlig na h-Alba
bosanski
galego





